Although pembrolizumab was associated with a trend toward improved survival in other subgroups, the benefit did not reach the prespecified threshold for statistical significance. Pembrolizumab was associated with a statistically significant 27% improvement in OS in the subgroup of patients with high PD-L1 expression, defined as CPS ≥10 (HR, 0.73 p =.0093) ( Table 2). SABCS Highlights from SABCS 2020: New data on CDK4/6 inhibitors, Highlights from SABCS 2021: T-DXd, KEYNOTE-355, PADA-1 Experts Discuss Clinical. The co-primary endpoints were overall survival (OS) and progression-free survival (PFS) in 3 patient cohorts defined by PD-L1 expression: CPS ≥10 ( n = 323), CPS ≥1 ( n = 636), and the intent-to-treat (ITT) study population ( N = 847). Patients were randomly assigned 2:1 to receive pembrolizumab every 3 weeks ( n = 566) or placebo ( n = 281) in addition to nab-paclitaxel, paclitaxel, or gemcitabine/carboplatin. Tumor samples were graded for PD-L1 expression based on the combined positive score (CPS), which measures PD-L1 expression on tumor cells, lymphocytes, and macrophages. The phase III KEYNOTE-355 trial enrolled 847 patients with locally recurrent inoperable or metastatic TNBC that has not been previously treated with chemotherapy in the advanced setting. Javier Cortes, M.D., Ph.D., of the International Breast Cancer Center, presented results from KEYNOTE-355 demonstrating the optimal PD-L1 expression threshold for pembrolizumab benefit in advanced or metastatic TNBC 2. SABCS 2022: HER2-low and HER2-zero triple-negative breast cancer show distinct biological differences. KEYNOTE-355: Pembrolizumab in Locally Advanced or Metastatic TNBC Merck has announced positive results from the Phase 3 KEYNOTE-355 trial investigating pembrolizumab in combination with chemotherapy as first-line treat. The benefit of pembrolizumab was consistent across subgroups, including those defined by disease stage and nodal status. Pembrolizumab added to neoadjuvant and adjuvant therapy was associated with a statistically significant and clinically meaningful 37% improvement in EFS (HR, 0.63 p =.00031) ( Table 1). 
The co-primary endpoints were pathologic complete response and EFS. Following surgery, patients received 9 additional cycles of adjuvant pembrolizumab or placebo. 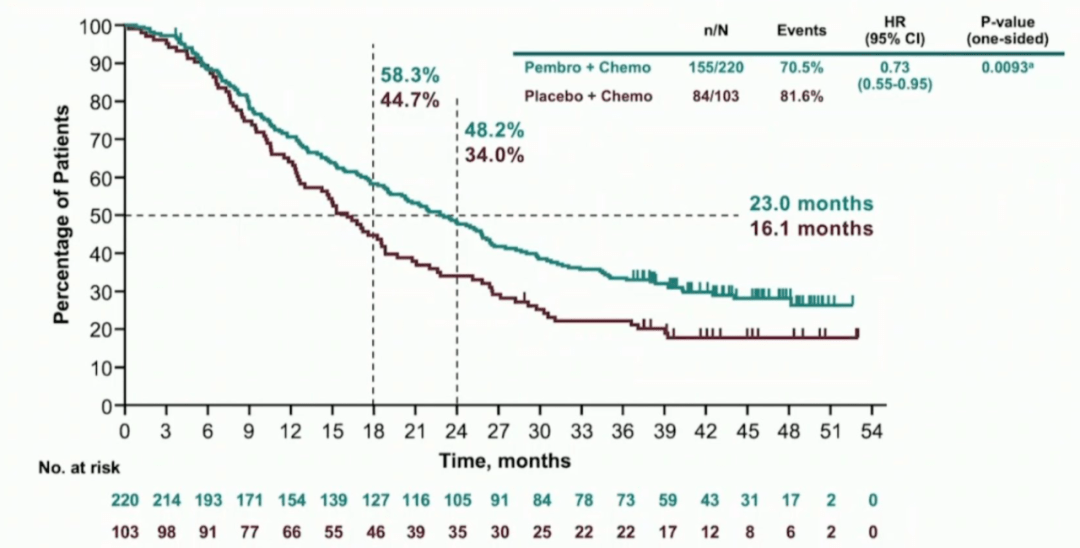
Patients were randomly assigned 2:1 to receive both neoadjuvant and adjuvant therapy with pembrolizumab 200mg every 3 weeks ( n = 784) or placebo ( n = 390).ĭuring the neoadjuvant phase of the trial, patients received pembrolizumab or placebo in addition to chemotherapy with carboplatin plus paclitaxel for 12 weeks, followed by doxorubicin or epirubicin plus cyclophosphamide for an additional 12 weeks. The phase III KEYNOTE-522 trial enrolled 1,174 patients with newly diagnosed, operable, stage II-III TNBC. Peter Schmid, M.D., Ph.D., of Barts Cancer Institute, presented findings from KEYNOTE-522 showing a 37% improvement in event-free survival (EFS) associated with neoadjuvant and adjuvant pembrolizumab in early-stage TNBC 1. KEYNOTE-522: Pembrolizumab in Early-Stage TNBC 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
